Abstract
Background: Wharton's Jelly Mesenchymal Stem Cells (WJ-MSC) are pluripotent stromal cells derived from umbilical cords, under development for several cell therapeutic applications. In vitro, they exert at least similar immune suppressive properties as bone marrow (BM)- or adipose tissue (AT)-derived MSCs. They have the advantage to be expanded with a higher proliferation rate in comparison to BM and AT-MSCs. By contrast with those latter, the optimal conditions and the main mechanisms of immunosuppression of WJ-MSCs have not been clearly defined. Here, we explored their mechanisms of action in controlling the proliferation of activated T cells in vitro and defined the best strategy for using them to control Graft Versus Host Disease (GVHD) in a pre-clinical mouse model of xeno-GVHD.
Methods: WJ-MSCs produced at clinical grade in our cell therapy unit primed or not with IFN-γ were characterized in terms of expression or production of immunosuppressive molecules. We then explored the potential pathways involved in their capacity to inhibit the proliferation and modulate cell metabolism of human T cells activated by anti-CD3/CD28 monoclonal antibodies. We finally defined the optimal conditions to control GVHD in NSG mice transplanted with human PBMCs.
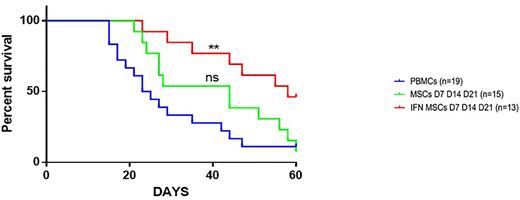
Results: WJ-MSCs could effectively inhibit the proliferation of activated T cells with a variable intensity and pre-incubation of WJ-MSCs with IFN-γ (IFN-WJ-MSC) for 48 h increased and homogenized their anti-proliferative capacities in vitro. After exploring different mechanisms of immunosuppression, we concluded that the major pathway used by WJ-MSCs to inhibit T cell proliferation was related to an increase in indoleamine2,3-dioxygenase (IDO) activity. IFN-WJ-MSCs induced a deeper depletion in tryptophan in the medium and increased the concentrations of kynurenine and kynurenic acid. However, a contact between LTs and IFN-WJ-MSCs was necessary to prevent T cell proliferation, independently of IDO activity. We observed that independently of IFN-γ activation and of IDO, WJ-MSCs can transfer mitochondrial material to activated T cells. This transfer was contact-dependent, as was the immunosuppressive action of WJ-MSCs. After mitochondrial transfer, activated T cells having received mitochondria proliferate significantly less than those that had not received mitochondria. Analysis of activated T cell metabolic pathways after co-culture with WJ-MSCs showed a modulation of T cell metabolism characterized by a decrease in aerobic glycolysis and an increase in oxidative phosphorylation. This phenomenon was partially reversed by IDO inhibition or by cell contact prevention. Inhibition of both IDO activity and cell contact completely restored the metabolism of activated T cells. Finally, we showed, in the NSG mouse model of xeno-GVHD, that IFN-γ-primed WJ-MSCs administrated sequentially at D7, D14 and D21 post-transplantation represented the best condition allowing to reduce clinical and histological signs of GVHD and increase survival (Figure 1). We explored the homing of WJ-MSCs and observed their presence in the lung and in the skin 24 hours after administration, then they became undetectable suggesting that their contact with T cells is nevertheless transiently possible in these organs after each administration.
Conclusion: Altogether, our results suggest for the first time that IFN-WJ-MSC inhibit LT activation via synergistic mechanisms involving IDO production and mitochondrial transfer modulating T cell metabolism. The results of the preclinical study led us to develop of a phase I clinical trial of several administrations of WJ-MSC for the prevention of GvHD.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal